PDF) Efficacy and Safety of Nebulized Formoterol as Add-on Therapy in COPD Patients Receiving Maintenance Tiotropium Bromide | Nicola Hanania - Academia.edu

Ipratropium Bromide 0.5mg and Albuterol Sulfate 2.5mg (Generic for DuoNeb)- Individually Foil Packed - 30/Box - Medical Warehouse

Comparison of the performance of inhalation nebulizer solution and suspension delivered with active and passive vibrating-mesh device - ScienceDirect

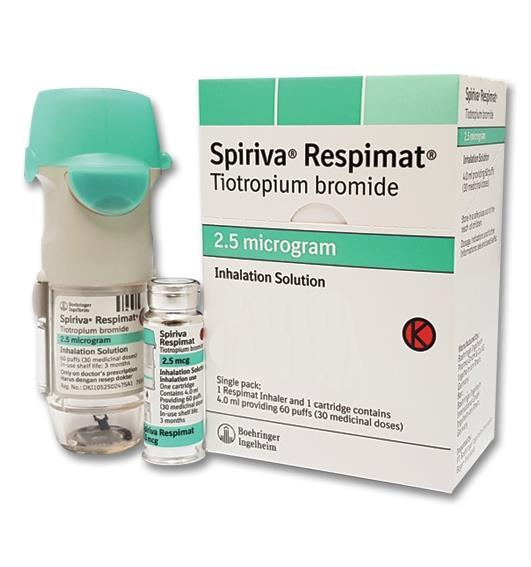
Spiriva Respimat 2.5 microgram, inhalation solution - Summary of Product Characteristics (SmPC) - (emc)

Ipratropium Bromide 0.5 mg and Albuterol Sulfate 3 mg Inhalation Solutions | Nephron Pharmaceuticals
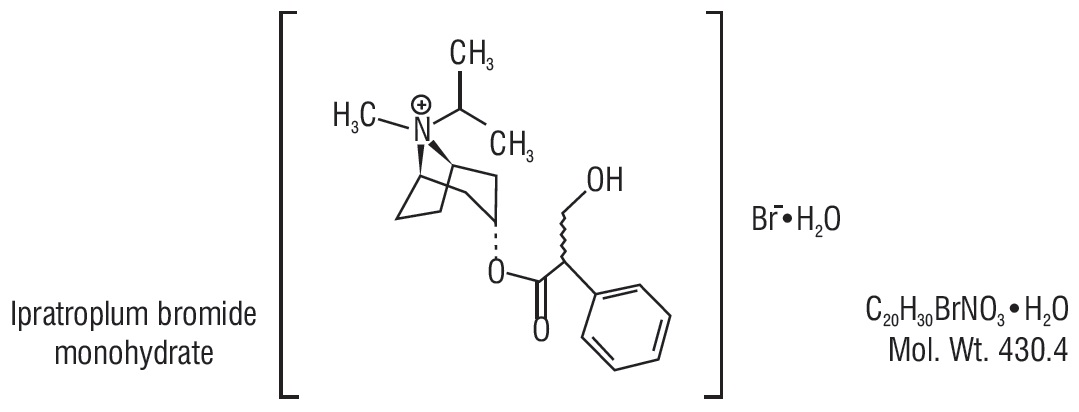
Ipratropium Bromide Inhalation Solution, USP 0.02% Rx only PRESCRIBING INFORMATION FOR INHALATION USE ONLY–NOT FOR INJECTION.
Deposition of revefenacin via nebulizer and tiotropium via DPI in the... | Download Scientific Diagram
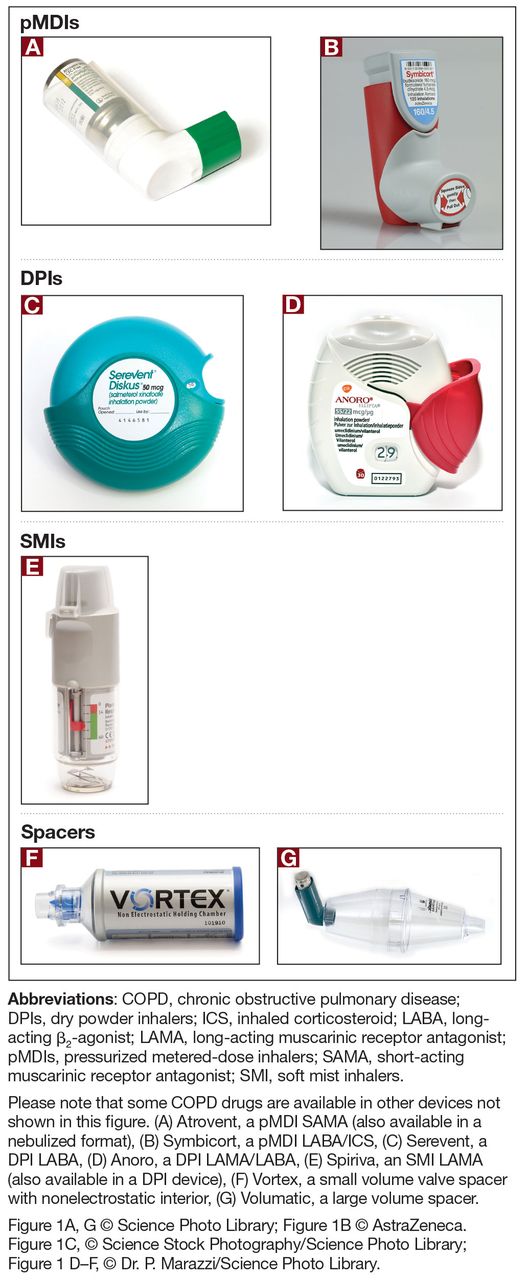
Considerations for Optimal Inhaler Device Selection in Chronic Obstructive Pulmonary Disease | Cleveland Clinic Journal of Medicine

Financial effect of converting ipratropium-albuterol therapy from inhalers to nebulizer treatments at an academic health system. | Semantic Scholar