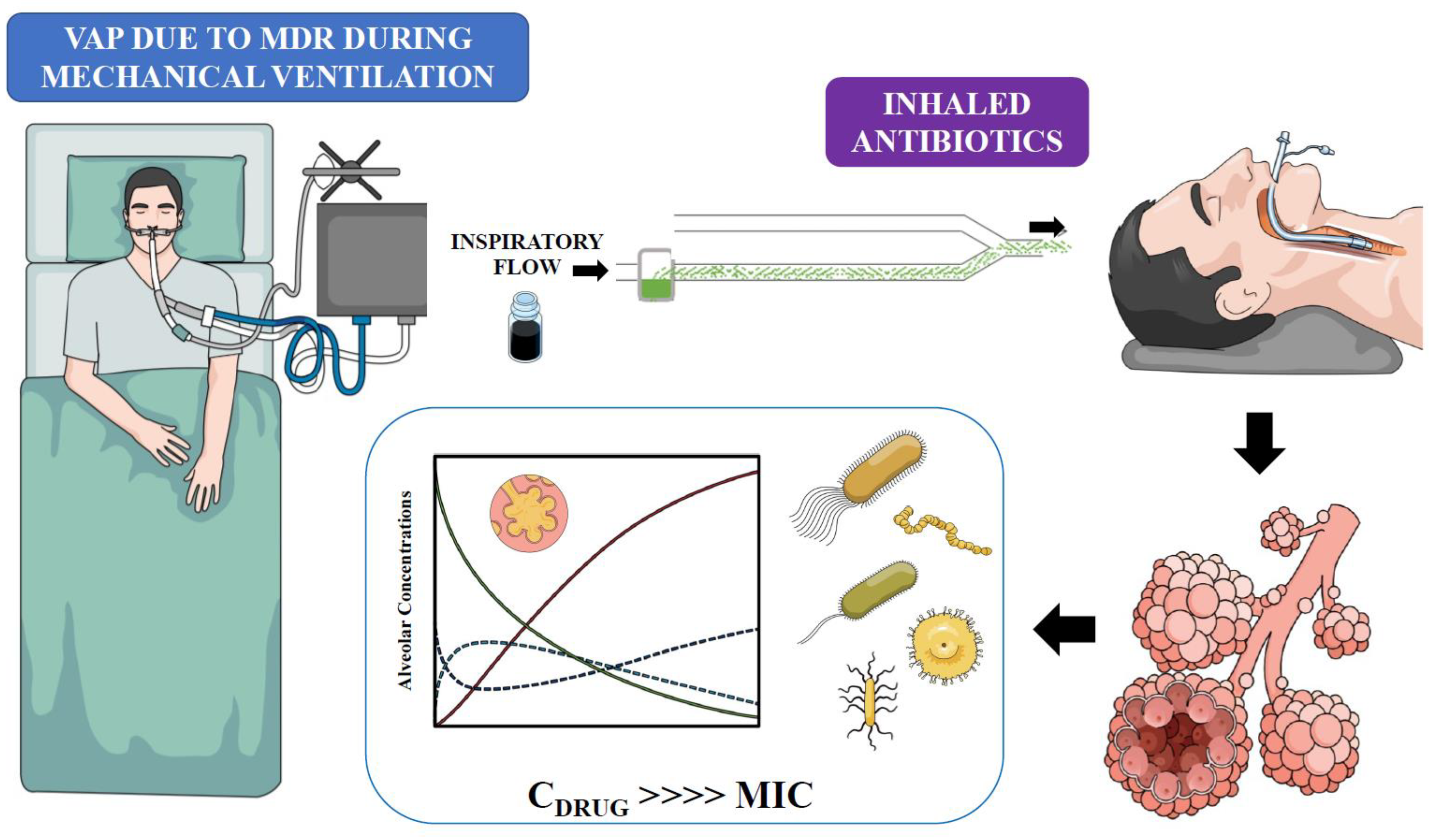
PDF) IN VITRO ASSESSMENT OF AEROSOLIZED AMIKACIN LUNG DOSE DELIVERED BY NKTR-061 PDDS CLINICAL DURING ON-VENTILATOR AND OFF-VENTILATOR USE

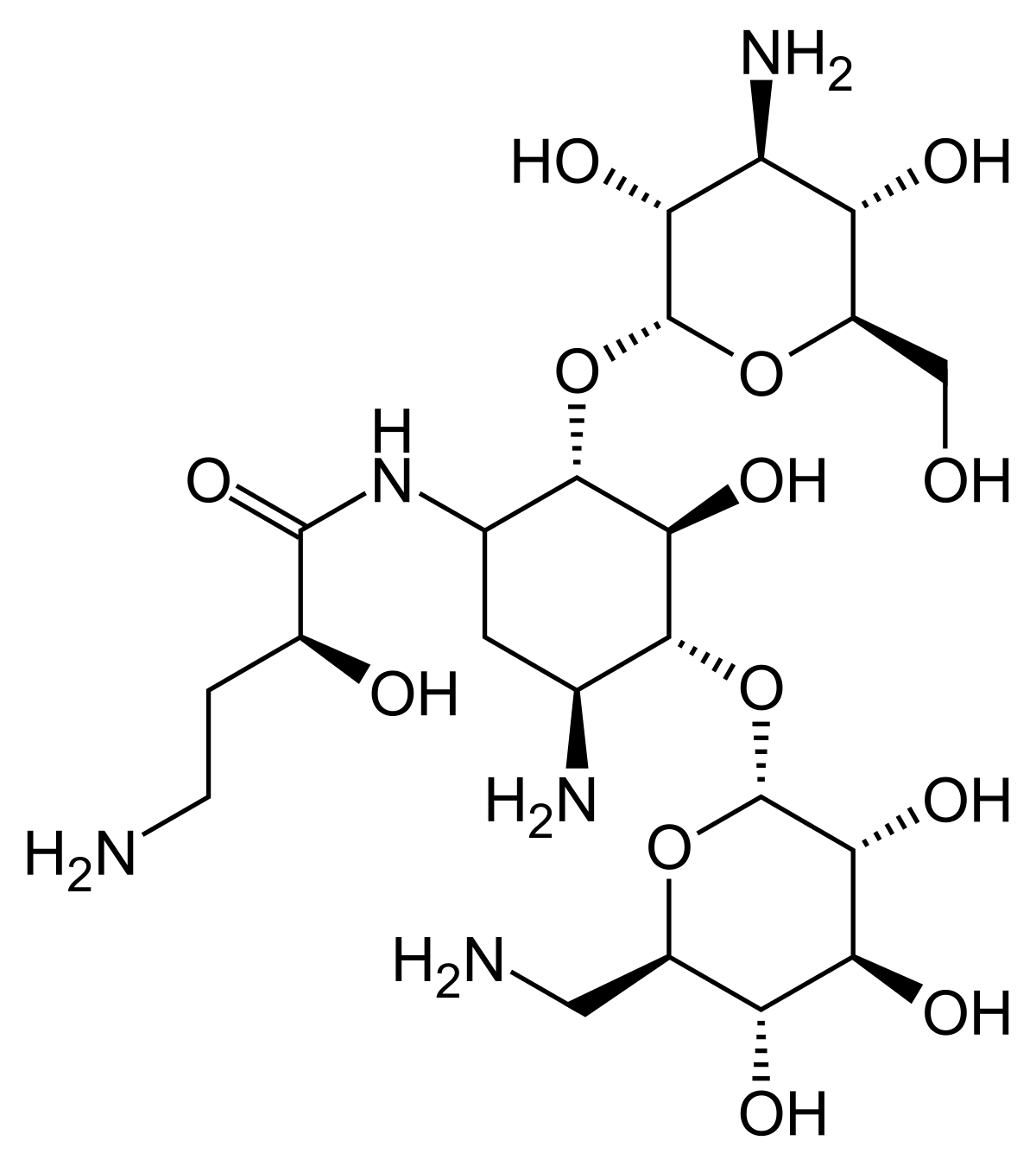
Colistin- and amikacin-loaded lipid-based drug delivery systems for resistant gram-negative lung and wound bacterial infections - ScienceDirect

These highlights do not include all the information needed to use ARIKAYCE safely and effectively. See full prescribing information for ARIKAYCE. ARIKAYCE® (amikacin liposome inhalation suspension), for oral inhalation use Initial U.S.

Robustness of aerosol delivery of amikacin liposome inhalation suspension using the PARI eFlow® Technology - Medicine

Pharmacokinetics of high-dose nebulized amikacin in mechanically ventilated healthy subjects | Intensive Care Medicine

Mean cumulative systemic absorption of amikacin as a function of time.... | Download Scientific Diagram
This handout provides distribution information for ARIKAYCE® (amikacin liposome inhalation suspension).